ПОТЕНЦИАЛ КАРРАГИНАНА ДЛЯ ОТКРЫТИЯ ПРЕПАРАТА ОТ COVID-19 ЧЕРЕЗ МОЛЕКУЛЯРНЫЙ ДОКИНГ С ИНГИБИТОРОМ АНГИОТЕНЗИНПРЕВРАЩАЮЩЕГО ФЕРМЕНТА 2 (ACE2) И ОСНОВНОЙ ПРОТЕАЗОЙ (MPRO) SAR-COV-2
DOI: https:/doi.org/ 10.18454/jbg.2022.18.2.001
Thet Htwe Aung *
Mawlamyine University, Mawlamyine, Myanmar
* Correspodning author (thethtweaung101[at]gmail.com)
Received: 22.10.2022; Accepted: 16.11.2022; Published: 22.11.2022
THE POTENTIAL OF CARRAGEENAN FOR THE DRUG DISCOVERY OF COVID-19 VIA MOLECULAR DOCKING WITH ANGIOTENSIN-CONVERTING ENZYME 2 (ACE2) AND THE MAIN PROTEASE (MPRO) OF SAR-COV-2
Research article
Abstract
The World Health Organization (WHO) has classified COVID-19 as a pandemic infection due to the global spread of new corona virus infections. Due to this virus infection, millions of people all around the world had to die or endure severe disease. It will be crucial to find new therapeutic treatments in order to prepare for a similar viral pandemic in the future. Carrageenans have apparently been effective against 12 viruses, including SAR-COV-2. In this investigation, angiotensin-converting enzyme 2 (ACE2) and main protease (Mpro) were used as molecular targets for virtual screening of kappa-, lambda-, and iota- carrageenans. When compared to antiviral drugs, the results show that all three carrageenans have substantial binding affinity for ACE2 and Mpro. The binding affinity of iota-carrageenan is greater than that of other compounds. The binding affinity suggests that carrageenans could be utilized to produce potent antiviral drugs.
Keywords: Covid-19, Carrageenan, binding affinity, ACE2, Mpro.
Тет Хтве Аунг *
Университет Моламьяйна, Моламьяйн, Мьянма
* Корреспондирующий автор (thethtweaung101[at]gmail.com)
Получена: 22.10.2022; Принята: 16.11.2022; Опубликована: 22.11.2022
ПОТЕНЦИАЛ КАРРАГИНАНА ДЛЯ ОТКРЫТИЯ ПРЕПАРАТА ОТ COVID-19 ЧЕРЕЗ МОЛЕКУЛЯРНЫЙ ДОКИНГ С ИНГИБИТОРОМ АНГИОТЕНЗИНПРЕВРАЩАЮЩЕГО ФЕРМЕНТА 2 (ACE2) И ОСНОВНОЙ ПРОТЕАЗОЙ (MPRO) SAR-COV-2
Научная статья
Аннотация
Всемирная организация здравоохранения (ВОЗ) классифицировала COVID-19 как пандемическую инфекцию в связи с глобальным распространением новых случаев заражения коронавирусом. Из-за этой инфекции миллионы людей по всему миру умерли или перенесли тяжелые заболевания. Крайне важно найти новые методы лечения, чтобы подготовиться к подобной вирусной пандемии в будущем. Каррагинаны оказались эффективны против 12 вирусов, включая SAR-COV-2. В данном исследовании ангиотензинпревращающий фермент 2 (ACE2) и основная протеаза (Mpro) были использованы в качестве молекулярных мишеней для виртуального скрининга каппа-, лямбда- и йота-каррагинанов. При сравнении с противовирусными препаратами результаты показали, что все три каррагинана обладают значительным вяжущим свойством к ACE2 и Mpro. Связывающая аффинность йота-каррагинана выше, чем у других соединений. Связывающая аффинность позволяет предположить, что каррагинаны могут быть использованы для производства мощных противовирусных препаратов.
Ключевые слова: Covid-19, каррагинан, связывающая аффинность, ACE2, Mpro.
1. Introduction
Carrageenans are linear polysaccharides that have been sulfated in various positions and are composed of alternating (1→3)-β-Dgalactopyranoses and (1→4)-α-D-galactopyranoses (or 3,6-anhydrogalactopyranoses).1 Due to its emulsifying, stabilizing, thickening, and gelling qualities, carrageenan is frequently employed as an emulsifier, stabilizer, thickener, and gelling ingredient in topical goods, cosmetics, and food preparations. According to their structural features, sulfation patterns, and the presence or absence of 3,6-anhydro bridges in α-linked galactose residues, they are divided into distinct categories. Among them, kappa-, lambda-, and iota- carrageenans are the three most economically used carrageenans. Lambda-carrageenans are formed by galactose units, whereas iota- and kappa- contain equal amounts of galactose and 3,6 anhydrogalactose. Kappa-carrageenan contains one sulfate group, iota-carrageenan contains two per disaccharide at axial positions, and lambda- has close to three equatorial sulfates.1 Red macroalgae such as Chondrous crispus, Kappaphycus alvarezii (Eucheuma cottonii), and E. denticulatum (E. spinosum) are the primary producers of carrageenan. Depending on the species and sometimes even the life stages of the same species, carrageenan structures can change [1].
As a biomolecule, carrageenan has many biological properties, including those of an antioxidant, an antibacterial, an anticoagulant, and an immunomodulator. Carrageenan also possesses a wide range of antiviral qualities. Twelve viruses (SARS-CoV-2, HSV, InfV, hRV, HIV, hCV, hCoVOC43, HPV, TMV, DENV, JEV, and RVFV) were reportedly combated by carrageenans [2].
Nasal sprays, throat sprays and lozenges containing carrageenans are approved common cold prevention options and have been launched in more than 20 countries.3 These products also have the potential of first line defense to inhibit the infection and transmission of SARS-COV-2 because carrageenans inhibit the viral infection of SARS-COV-2 Wuhan type in a similar way as the Alpha, Beta, Gamma, and Delta variants.3 The most commonly used carrageenan in these products is iota-carrageenan since iota-carrangeenan displayed an at least 10-fold higher efficacy when compared to lambda- and kappa-carrageenan [3].
A severe case of a respiratory illness was reported in Wuhan, Hubei People's Republic of China, during the end of December 2019.4 Due of its genetic similarities to an earlier known coronavirus (SARS-CoV), 2019-nCoV was eventually renamed as SARS-CoV-2 by the International Committee on Taxonomy of Viruses (ICTV). The World Health Organization (WHO) proclaimed Coronavirus Disease-19 (COVID-19), a coronavirus-induced disease, to be a pandemic on March 11, 2020 as a result of its extensive global spread. SARS-COV-2 Omicron B.1.1.529 variant was initially reported to the World Health Organization (WHO) on 24 November 2021 from South Africa.4 Around the world, Coronavirus Disease-19 has been linked to about 128 million confirmed illnesses and more than 2.8 million fatalities as of 30 March 2021 [4].
Although there are already effective COVID-19 vaccines and drugs that can be used to prevent or treat COVID-19, viral variants can provide significant obstacles for these drugs and vaccines. Furthermore, the WHO believes that a future viral pandemic of a similar type is inevitable [5]. Therefore, it is essential to keep working on discovering new drugs that may be used in the early stages of COVID-19 to stop progression.
The angiotensin-converting enzyme 2 (ACE2) receptor, which is expressed in a number of human organs, serves as a pathway for both SARS-CoV-2 and SARS-CoV to enter host cells [12]. Mpro (also known as 3CLpro) protease is regarded as a desirable protein target in COVID-19 drug development, particularly for the screening of substances that might prevent coronavirus replication. More than 95% of the SARS-CoV-2 protease's composition is identical to the SARS-CoV-1 Mpro [11].
In this investigation, two enzymes were used as molecular targets against the coronavirus: Mpro from the virus-cell and ACE2 receptor from the host cell. Numerous structural and non-structural proteins are encoded by the (+) SS RNA virus known as SARS-CoV-2. The Mpro is a non-structural protein that divides two replicase polyproteins into mature proteins needed to enable viral transcription and replication. In this way, we can prevent the spread of the virus by inhibiting Mpro, while tiny molecules that inhibit ACE2's catalytic pocket can alter ACE2's conformation and prevent SARS-CoV-2 from entering host cells by ACE2. In order to prevent virus replication, we chose Mpro, protease as a target, and ACE2 receptor to prevent SARS-CoV-2 entry [10]. Remdesivir and Favipiravir, two widely used antiviral drugs, were utilized in this work as positive controls to compare the binding affinities to the target proteins ACE2 and Mpro.
According to the findings, all three carrageenans have the ability to operate as bioactive substances that can protect and treat Covid-19. Iota-carrageenan would be the most effective of these for both treating and defending Covid-19. Carrageenans may be used in future research and development of antiviral agents to combat the Coronavirus. Carrageenans are on the FDA list of substances that are generally recognized as safe for consumption, thus additional ADME (absorption, distribution, metabolism, and excretion) research is not necessary to approve them.
2. Methods
2.1. Selection of ligands and protein
The SDF format of two antiviral compounds, Remdesivir and Favipiravir, and three carrageenan compounds, kappa-, lambda-, and iota-carrageenan, with CHEBI numbers of 10583, 37167, and 37168, were obtained from Chemical Entities of Biological Interest (ChEBI) (https://www.ebi.ac.uk/chebi). The 3D structure of the main protease (Mpro) and angiotensin-converting enzyme 2 (ACE2) was obtained using PDB IDs 6m0j and 6lu7 from protein data bank (http://www.rcsb.org).
2.2. Molecular Docking study
The CB-Dock web server (http://cao.labshare.cn/cb-dock/) [6] was used to analyze the docking of the compounds. The CB-Dock online server simply requires a protein file in PDB format and a ligand file in MOL2, MOL, or SDF format as input. Following submission, the CB-Dock examines the input files and uses OpenBabel and MG Tools to convert them to pdbqt formatted files. CB-Dock then predicts the protein's cavities and determines the centres and sizes of the top N (by default, n=5) cavities. AutoDock Vina is used to dock the centres and sizes, as well as the pdbqt files. After computing, the final findings are displayed. In the table, users can look up binding scores, cavity size, and docking parameters for projected binding modes. However, in this experiment, only the optimal pose of binding scores (vina scores) for each molecule was assessed, as indicated in Table 1.
2.3. Protein-ligand interaction
The best poses of the protein-ligand complex pdb file acquired from the CB-Dock web server were submitted to the The ProteinPlus web server (http://protein.plus). PoseView [7] was used to analyze the 2D interaction diagram with hydrogen and hydrophobic contacts for the protein-ligand interaction.
3. Results and Discussion
In order to estimate the binding sites and affinities with the Autodock Vina program, carrageenans were molecularly docked with HER2 and Mpro utilizing CB- Dock, an online application. Since 2019, the CB- Dock web server, which Autodock Vina is utilized for, has seen roughly 200 entries per day posted by academics from around the world. AutoDock Vina is an open-source, free software program that quickly determines the binding affinity (Vina scores) that roughly represent the binding energy (Kcal/mol).
Affinity indicates the extent of the drug's interaction with the receptor. A low vina score denotes a high binding affinity of the protein to the ligand. Drug candidates are picked from ligands that bind firmly to the target protein because the stronger the connections, the more the ligand will influence the physiological function of the target proteins. A high affinity usually results in a reduced dose requirement [8].
Lambda- and iota-carrageenan had the lowest scores for ACE2 with vina values of -8.5, followed by kappa-carrageenan (-8.4), albeit their scores are extremely close to one another. Remdesivir and Favipiravir, the standard antiviral drugs, showed vina scores of (-8.5) and (-6.4) for ACE2. Iota-carrageenan had the lowest vina score for Mpro, with a value of -7.7, followed by Kappa-carrageenan (-7.3), and lambda-carrageenan (-7.3). (-6.9). Vina scores of (-7.5) and (-5.8) were displayed for Mpro by Remdesivir and Favipiravir (table 1).
All three carrageenans displayed the same binding affinity as Remdesivir for the ACE2 protein target, but they all have better binding affinities than Favipiravir. Both kappa- and iota-carrageenan and Remdesivir had the same binding affinity for Mpro, whereas lambda-carrageenan had a little lower affinity. However, compared to Favipiravir, they all showed a higher binding affinity. Carrageenans demonstrated a higher binding affinity for the ACE2 protein target than Mpro. Iota-carrageenan was the carrageenan with the highest binding affinity for both ACE2 and Mpro.
For structural bioinformatics, pharmaceutical development, and biological research, it is crucial to characterize interactions in protein-ligand complexes. It was projected that kappa-carrageenan would bind ACE2 through three H-bonds with Tyr196A, Tyr566A, and Lys562A (figure 1) and Mpro through three H-bonds with Asn203A, Ser158A, and Gln110A (figure 4). Lambda-carrageenan was projected to bind ACE2 through six H-bonding interactions with Asp382A, Asn394A, Arg514A, Glu398A, Lys562A, and Trp203A (figure 2) and Mpro through five H-bonding interactions with Arg105A, Gln110A, Glu240A, Lys102A, and Thr243A (figure 5). Iota-carrageenan was expected to bind ACE2 through four H-bonding with Arg514A, Asp206A, Gln102A, and Tyr385A (figure 3) and to bind Mpro through seven H-bonding with Asp153A, Gln110A, Lys102A, Phe294A, Thr111A, Thr292A, and Ser158A (figure 6). Most high-affinity ligands require the strong hydrogen bonds [9]. It follows that Lambda- and Iota-carrageenan are ligands with high affinity for ACE2 and Mpro.
Table 1 – Binding parameters between ligands and target proteins
|
Compound names |
ACE2 |
Mpro |
||
|
Vina score |
Amino acids involved in hydrogen bonds |
Vina score |
Amino acids involved in hydrogen bonds |
|
|
Kappa- carrageenan |
-8,4 |
Tyr196A, Tyr566A, Lys562A |
-7,3 |
Asn203A, Ser158A, Gln110A |
|
lambda- carrageenan |
-8,5 |
Asp382A, Asn394A, Arg514A, Glu398A, Lys562A, Trp203A |
-6,9 |
Arg105A, Gln110A, Glu240A, Lys102A, Thr243A |
|
iota- carrageenan |
-8,5 |
Arg514A, Asp206A, Gln102A, Tyr385A |
-7,7 |
Asp153A, Gln110A, Lys102A, Phe294A, Thr111A, Thr292A, Ser158A |
|
Remdesivir |
-8,5 |
Arg403, Asn33A, Gln96A, Tyr453E, Tyr505E |
-7,5 |
Arg105A, Gln110A, Thr111A |
|
Favipiravir |
-6,4 |
Gly496E |
-5,8 |
Asn151A, Asp295A, Gln110A, Thr292A |
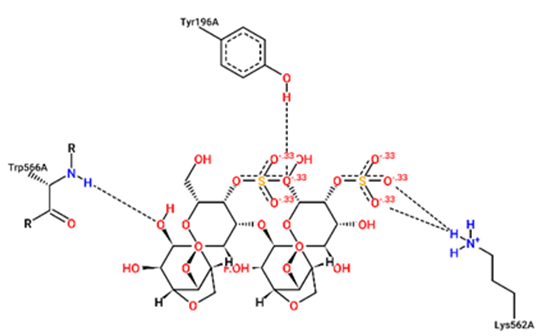
Fig. 1 – 2D protein- ligand interaction diagrams obtained by ProteinPlus at the active site of target proteins
ACE2 for kappa-carrageenan
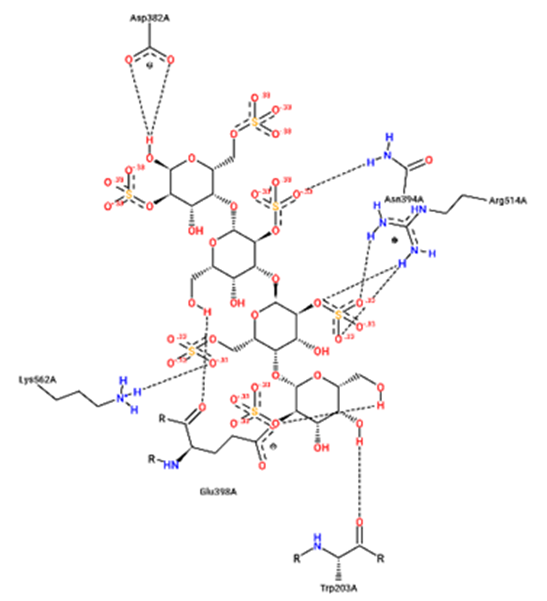
Fig. 2 – 2D protein- ligand interaction diagrams obtained by ProteinPlus at the active site
of target proteins ACE2 for lambda- carrageenan
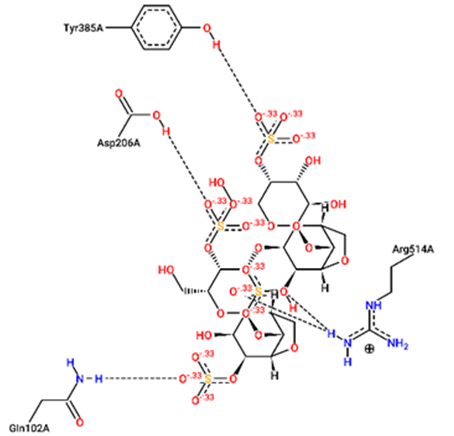
Fig. 3 – 2D protein- ligand interaction diagrams obtained by ProteinPlus at the active site
of target proteins ACE2 for iota- carrageenan
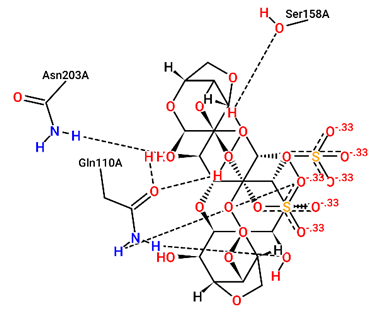
Fig. 4 – 2D protein- ligand interaction diagrams obtained by ProteinPlus at the active site
of target proteins Mpro for kappa- carrageenan
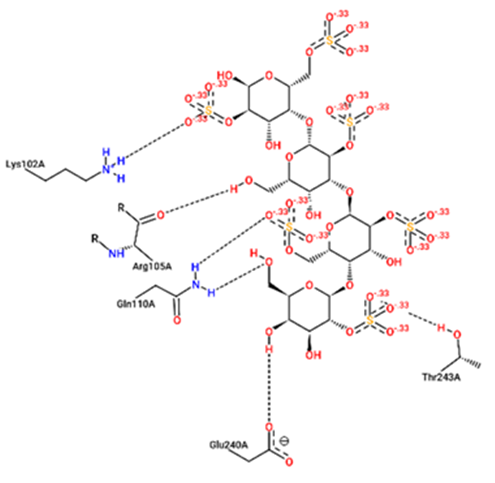
Fig. 5 – 2D protein- ligand interaction diagrams obtained by ProteinPlus at the active site
of target proteins Mpro for lambda- carrageenan
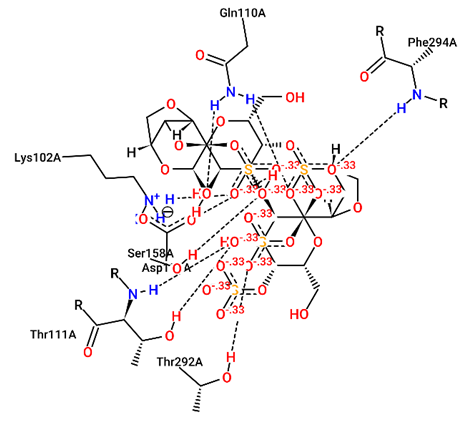
Fig. 6 – 2D protein- ligand interaction diagrams obtained by ProteinPlus at the active site
of target proteins Mpro for iota- carrageenan
|
Conflict of Interest |
Конфликт интересов |
|
None declared. |
Не указан. |
Список литературы
Alvarez-Vinas M. Antiviral Activity of Carrageenans and Processing Implications / M. Alvarez-Vinas, S. Souto, N. Florez-Fernandez et al. // Mar. Drugs. 2021, 19, 437. DOI: 10.3390/md19080437
Padmi H. Macroalgae Bioactive Compounds for the potential Antiviral of SARS-CoV-2: An In Silico Study / H. Padmi, V.D. Kharisma, A.N.M. Ansori et al. // J Pure Appl Microbiol. 2022; 16(2): 1018-1027. DOI: 10.2227/JPAM.16.2.26
Froba M. Iota- Carrageenan Inhibits Replication of SARS-CoV-2 and the Respective Variants of Concern Alpha, Beta, Gamma and Delta / M. Froba, M. GroBe, C. Setz et al. // Int. J. Mol. Sci. 2021, 22, 13202. DOI: 10.3390/ijms222413202
Yadav R. Role of structural and Non-structural Proteins and Therapeutic Targets of SARS-CoV-2 for COVID-19 / R. Yadav, J.K. Chaudhary, N. Jain et al. // Cells. 2021, 10, 821. DOI: 10.3390/cells10040821
Robinson B.W.S. Why we still need drugs for COVID-19 and can’t just rely on vaccines / B.W.S. Robinson, A. Tai, K. Springer // Respirology. 2022; 27: 109-11. DOI: 10.1111/resp.14199
Liu Y. CB- Dock2: improved protein- ligand blind docking by integrating cavity detection, docking and homologous template fitting / Y. Liu, X. Yang, J. Gan et al. // Nucleic Acids Research, 2022, 50, Web Service Issue. W159-W164. DOI: 10.1093/nar/gkac394
Stierand. Poseview- molecular interaction patterns at a glance / Stierand and Rarey // Journal of Cheminformatics. 2010. 2(Suppl 1): P50. DOI: 10.1186/1758-2946-2-S1-P50
Aung T.H. Proximicin A-C as prospective HER2- positive and negative breast cancer drugs: Molecular docking and in silico ADME modeling / T.H. Aung // IPS Journal of Molecular Docking Simulations, 2022, 1(1), 1-11. DOI: 10.54117/ijmds.v1i1.9
Pantsar T. Binding Affinity via Docking: Fact and Fiction / T. Pantsar, A. Poso // Molecules. 2018, 23, 1899; DOI: 10.3390/molecules2381899
Joshi T. European Review for Medical and Pharmacological Sciences / T. Joshi, T. Joshi, P. Sharma, et al. 2020, 24: 4529-4536.
Vicidomini C. In silico investigation on the interaction of Chiral phytochemicals from Opuntia ficus-indica with SARS- CoV-2 Mpro / C. Vicidomini, V. Roviello, G.N. Roviello // Symmetry. 2021, 13, 1041. DOI: 10.3390/sym1361041
Ni W. Role of angiotensin- converting enzyme 2 (ACE2) in Covid 19 / W. Ni, X. Yang, D. Yang et al. // Crit Care. 24, 2020, 422. DOI: 10.1186/s13054-020-03120-0
Список литературы
Alvarez-Vinas M. Antiviral Activity of Carrageenans and Processing Implications / M. Alvarez-Vinas, S. Souto, N. Florez-Fernandez et al. // Mar. Drugs. 2021, 19, 437. DOI: 10.3390/md19080437
Padmi H. Macroalgae Bioactive Compounds for the potential Antiviral of SARS-CoV-2: An In Silico Study / H. Padmi, V.D. Kharisma, A.N.M. Ansori et al. // J Pure Appl Microbiol. 2022; 16(2): 1018-1027. DOI: 10.2227/JPAM.16.2.26
Froba M. Iota- Carrageenan Inhibits Replication of SARS-CoV-2 and the Respective Variants of Concern Alpha, Beta, Gamma and Delta / M. Froba, M. GroBe, C. Setz et al. // Int. J. Mol. Sci. 2021, 22, 13202. DOI: 10.3390/ijms222413202
Yadav R. Role of structural and Non-structural Proteins and Therapeutic Targets of SARS-CoV-2 for COVID-19 / R. Yadav, J.K. Chaudhary, N. Jain et al. // Cells. 2021, 10, 821. DOI: 10.3390/cells10040821
Robinson B.W.S. Why we still need drugs for COVID-19 and can’t just rely on vaccines / B.W.S. Robinson, A. Tai, K. Springer // Respirology. 2022; 27: 109-11. DOI: 10.1111/resp.14199
Liu Y. CB- Dock2: improved protein- ligand blind docking by integrating cavity detection, docking and homologous template fitting / Y. Liu, X. Yang, J. Gan et al. // Nucleic Acids Research, 2022, 50, Web Service Issue. W159-W164. DOI: 10.1093/nar/gkac394
Stierand. Poseview- molecular interaction patterns at a glance / Stierand and Rarey // Journal of Cheminformatics. 2010. 2(Suppl 1): P50. DOI: 10.1186/1758-2946-2-S1-P50
Aung T.H. Proximicin A-C as prospective HER2- positive and negative breast cancer drugs: Molecular docking and in silico ADME modeling / T.H. Aung // IPS Journal of Molecular Docking Simulations, 2022, 1(1), 1-11. DOI: 10.54117/ijmds.v1i1.9
Pantsar T. Binding Affinity via Docking: Fact and Fiction / T. Pantsar, A. Poso // Molecules. 2018, 23, 1899; DOI: 10.3390/molecules2381899
Joshi T. European Review for Medical and Pharmacological Sciences / T. Joshi, T. Joshi, P. Sharma, et al. 2020, 24: 4529-4536.
Vicidomini C. In silico investigation on the interaction of Chiral phytochemicals from Opuntia ficus-indica with SARS- CoV-2 Mpro / C. Vicidomini, V. Roviello, G.N. Roviello // Symmetry. 2021, 13, 1041. DOI: 10.3390/sym1361041
Ni W. Role of angiotensin- converting enzyme 2 (ACE2) in Covid 19 / W. Ni, X. Yang, D. Yang et al. // Crit Care. 24, 2020, 422. DOI: 10.1186/s13054-020-03120-0
